Turkeydinner at Thanksgiving brings the family together, celebrates the bountiful harvest, and puts you to sleep just as the NFL games are ready to start. Many people think that if you eat less turkey and fill up on the other goodies you can escape the post-Thanksgiving meal sleepiness. Other people look forward to eating seconds and thirds and then stretching out on the couch for a long nap, forcing Aunt Ethel to sit in the chair with the spring that surprises you every once in a while.
The culprit, or the hero, in this eat and sleep saga is said to be the tryptophan in the turkey. Other people think that it is simply how much you eat, not the turkey's tryptophan, but it isn’t quite that simple. What is tryptophan, and is it indeed responsible for the snoring that follows Thanksgiving dinner? Some background will help.
Tryptophan is an amino acid, one of the twenty standard building blocks of proteins. Each amino acid has a similar basic structure, as shown in the picture below. The central carbon has an amino group (NH3) on one side and a carboxylic acid (COO-) moiety on the other; hence the name – amino acid. The third side group is a simple hydrogen (H), while the fourth side (R) refers to any of several different side groups and is what makes one amino acid different from one another.
Tryptophan is an aromatic amino acid, meaning that its side group contains a six-sided carbon ring structure (each corner represents a carbon). It also has a second ring group of four carbons and a nitrogen. As such, it is the largest and most massive of all the standard amino acids. However, tryptophan is the least abundant amino acid in plant and animal proteins; it accounts for only 1-1.5% of the total number of amino acids in proteins.
Tryptophan’slarge structure and intricate rings make it costly to produce in terms of ATP invested. In fact, it takes so much energy to make that we have stopped making tryptophan all together. Tryptophan is abundant in a number of food sources commonly available to humans, so over evolutionary time we have turned it into an essential amino acid. True, it is essential for life, but here the word “essential” means that we MUST get it from our diet, we cannot produce it ourselves.
Of the 20 standard amino acids, 10 are essential in humans (9 that we must eat and 1 that we make from an essential amino acid), but bacteria make them all just fine - although the parents of newborns may wish it wasn’t so. Gut bacteria make tryptophan or use the tryptophan we eat. They transform it into molecules they need to survive, but the byproducts of these reactions are skatole and indole – these are the precious little molecules that give dirty diapers that wonderful smell!
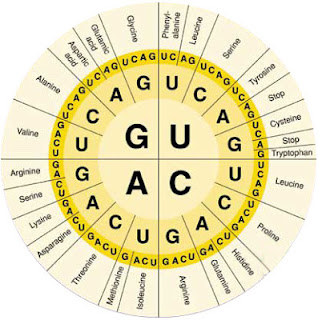
Tryptophan is different from many other amino acids in another way as well; it gets no respect from the genetic code. Each amino acid is coded for by a group of three RNA bases, together called a codon. Since there are four different bases in mRNAs (A, C, G, and U – remember that T is used in DNA but not RNA), then there are 64 different codons (4 x 4 x 4). This is more than the 20 amino acids that the codons code for, so most amino acids have two or three codons that signals that they should be added to the growing peptide. But tryptophan is encoded by only one codon (UGG).
It may make sense that an amino acid that is not used often in proteins might rate only one codon, but the amino acid methionine is used much more often than tryptophan, and it's only coded for by one codon as well (AUG). You know nature must have a reason why tryptophan has a single codon, we just don't know it yet.
Eventhough it is used sparingly in proteins, tryptophan is an essential amino acid - don’t eat enough of it and you die. This is because tryptophan’s most essential functions have nothing to do with protein synthesis or structure – tryptophan is important to your brain function. The crucial neurotransmitter, serotonin, is synthesized only from tryptophan.
It takes two enzymes to turn tryptophan into serotonin (also called 5-HT). First is tryptophan hydroxylase; hydroxylase means it splits water, here it adds an OH to tryptophan. Next, the amino acid decarboxylase removes a carboxylic acid (COOH), producing serotonin.
Amongst the many functions of serotonin are a few that are not brain related. Serotonin is released by enterochromaffin cells that line your gut to tell your gut to move. The movement helps push the food along your digestive tract, but serves a protective function.
If you eat something toxic, the enterochromaffin cells produce more serotonin – your gut moves much faster, and you get diarrhea. If even more serotonin is made and released, it moves through the bloodstream to your stomach and esophagus and causes you to vomit.
But it is in the CNS that serotonin has its significant activities. As a neurotransmitter, it is responsible for controlling how electric messages are passed from one neuron to another. When serotonin is released in the synapse (the gap between the upstream and downstream neurons) and is taken up by adjacent neurons, it produces a sense of well-being.
It isn’t surprising that depressed individuals often have low blood levels of tryptophan, as well as reduced serotonin. Classic treatments for depression include increased tryptophan intake, monoamine oxidase (MAO) inhibitors, and serotonin reuptake inhibitors (SSRI). With more tryptophan, you make more serotonin – problem solved. On the other hand, MAO’s break down serotonin, so their inhibitors enhance the action of tryptophan. SSRI’s prevent the reuptake, this leaves serotonin in the synapse longer. Both types of drugs make tryptophan more likely to be taken up by downstream neurons.
Unfortunate, but interesting, is the study showing that the suicidal thoughts that sometimes accompany anti-depressant therapies (TESI – treatment enhances suicidal ideation) use may be related to polymorphisms in one form of the tryptophan hydroxylase enzyme that starts the serotonin production from tryptophan.
When non-suicidal patients were compared to those with TESI or those who were suicidal without treatment, a pattern emerged. Only those with TESI showed a polymorphism pattern in the tryptophan hydroxlyase 2 (TPH2) gene. This polymorphism had previously been associated with suicide victims and major depressive disorder. It seems that a slight alteration in function of TPH2 due to a single nucleotide change can contribute to the genetic background of treatment induced suicidal thoughts.
The feeling of general well being induced by serotonin also participates in the sleep/wake cycle. So is tryptophan – through serotonin – responsible for the post-Thanksgiving nap? Well… yes and no, it's an accomplice in a larger conspiracy.
Serotonin is use to produce the hormone melatonin, and melatonin promotes sleep, so you could say turkey dinner promotes sleep. But turkey doesn’t have that much tryptophan! Tofu has much more tryptophan than turkey, but you don’t get a post-Chinese takeout urge to sleep, so what gives?
The melatonin effect has to do more with how much of everything else you eat at Thanksgiving dinner, especially carbohydrates. Here is how it works – eating lots of carbohydrates causes a release of insulin into the blood (to reduced blood glucose levels). Another function of insulin is to promote the uptake of some amino acids (but not tryptophan) into muscle cells. This leaves the blood higher in tryptophan as compared to other amino acids than it would normally be.
The brain takes in amino acids through a neutral amino acid transporter, which now finds more tryptophan than other neutral amino acids, so the brain level of tryptophan goes up. More tryptophan in the brain, more serotonin – more serotonin, more melatonin. More melatonin = nap time! So if you want to avoid the post-Thanksgiving nap, eat the turkey and skip the mashed potatoes.
You didn’t know how much tryptophan controlled your daily life, did you? Well, there’s more. Tryptophan is also important in synthesizing niacin, a.k.a. vitamin B3 or nicotinic acid. Niacin is important in production of NAD/NADH for energy metabolism, for production of steroid hormones and balance of lipid forms in the blood, and as an anti-convulsant.
The tryptophan-niacin connection is made stronger by recent evidence that high dietary tryptophan can prevent epileptic seizures in mice. In this study, a whey protein called alpha-lactoalbumin (ALAC) was found to have much tryptophan, much higher levels than in most proteins. Feeding epileptic mice ALAC resulted in reduced numbers of seizures.
So even if you don’t want to sleep or think happy thoughts, you still need to eat food that contain tryptophan or niacin. And many of those foods are plants, because plants use tryptophan to control their own activities. Tryptophan is easily converted to auxins, a type of plant hormone. Auxins are responsible for several different plant behaviors, namely the falling leaves in autumn and ripe fruits all year long.
Having dietary choices for tryptophan is good, and plants provide our major source. However, cooking grains and corn reduces usable tryptophan and niacin levels dramatically, so poorer environments where corn is the staple food need also to have additional dietary sources of tryptophan. A deficiency of this amino acid leads to some disturbing conditions. Low tryptophan leads to low serotonin levels and agitation, insomnia, and depression. A study in the Archives of General Psychiatry stated that chronically low levels of tryptophan led to relapses of purging behaviors in bulimics.
More amazingly, studies in the 1970’s to 1990’s suggest that low tryptophan levels can lead to increases in religious fanaticism. Several studies from a single author correlate the Aztec human sacrificial ceremonies to the times of year when their diets depended more on foods that had less tryptophan. Think of all the lives that could have been saved by tofu!
But turkey is more than just tryptophan. You have to love an animal that has caruncles, a wattle, and a snood!
For more information or classroom activities, see:
Genetic code –
Neurotransmitters –
http://science.education.nih.gov/supplements/nih2/addiction/activities/lesson2_neurotransmission.htm
http://science.education.nih.gov/supplements/nih2/addiction/activities/activities_toc.htm